Because corneal irregularities can compromise the accuracy of IOL power calculations, it is essential that corneal surgeons identify the causes of these irregularities and treat these causes prior to IOL power determination.
Here, I discuss the primary causes of irregular astigmatism, the related preoperative treatment choices, and the IOL options for each cause.
Causes of Irregular Astigmatism
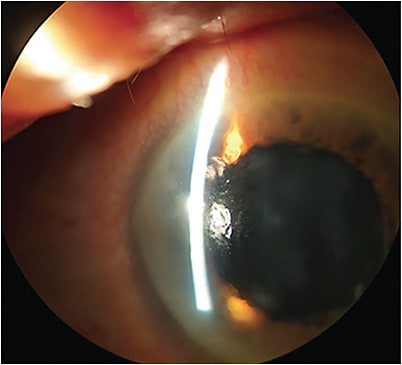
• Epithelial basement membrane dystrophy (EBMD). Patients who have EBMD present with sharply demarcated, faint grey-white opacities, as well as grey microcysts and fingerprint lines within the corneal epithelium. When the grey-white opacities are in the visual axis, they can induce irregular astigmatism.
Corneal topography aids in detecting subtle EBMD changes. Specifically, the axial map will show irregularity. The Placido ring image can be helpful. Areas of EBMD result in irregularity of the mires, which normally should be smooth and concentric.
• Keratoconus (KCN). KCN is an ectatic disease of the cornea with a range of clinical signs, depending on disease severity. In its early stages, diagnostic imaging is used to make the diagnosis. Inferior steepening is seen on corneal topography. Tomography can pick up earlier corneal changes, including posterior corneal elevation and paracentral corneal thinning. Later stages of disease that can be detected on clinical exam include stromal thinning and protrusion, Kayser-Fleisher rings, Vogt’s striae, and stromal scarring.
• Salzmann’s nodular degeneration (SND) and pterygium. SND appears as elevated grey-white lesions typically located in the mid-peripheral or peripheral cornea.
A pterygium appears as a mass of fibrovascular tissue that extends from the conjunctiva onto the corneal surface. Often, corneal flattening is noted on topography in the area of the nodule or head of the pterygium.
Despite their peripheral location, both SND and pterygia can induce visually significant corneal astigmatism. This astigmatism can be regular or irregular.

Related Preoperative Choices
• EBMD. Treatment of visually significant EBMD consists of epithelial debridement and removal of the irregular basement membrane, which can be performed with a 57 blade, a blunt Tooke corneal knife, or a 3.3 mm diameter diamond burr. Alternatively, phototherapeutic keratectomy (PTK) can be performed with the excimer laser to ablate 5 µm to 10 µm of Bowman’s membrane after epithelial debridement.1
The cornea is then given at least 6 weeks to 8 weeks to heal, at which point repeat IOL measurements are performed. The actual healing time can vary from patient to patient, and often a second set of IOL measurements are taken a week or two later to confirm stability.
If stability is not achieved, surgery would be deferred until repeat topography and measurements stabilize, which can take several months.
• KCN. Treatment of irregular astigmatism secondary to KCN is a challenge. The reason: The surgical interventions of corneal cross-linking (CXL), corneal ring segments, or corneal transplantation typically do not correct the significant amount of irregular astigmatism in the cornea.
The good news: Topography-guided PTK can treat this astigmatism and accompanying topographic irregularities. When treated in the context of impending cataract surgery, spherical correction is not needed, as this can be corrected with the IOL. Thus, this limits ablation depth in already-thin corneas.
To decrease the risk of postoperative ectasia, KCN patients can benefit from CXL combined with topography-guided PRK. Specifically, typically following CXL, the cornea is given between 3 months and 6 months to stabilize before topography-guided photorefractive keratectomy is performed. The cornea then needs at least 3 additional months to heal before IOL calculations can be acquired for cataract surgery. Several studies have demonstrated the safety and efficacy of this technique.2
• SND and pterygium. Significant changes in both the magnitude and orientation of the astigmatism can occur following surgical excision. SND is treated with superficial keratectomy. Adjuvant PTK can be used as well to further smooth out the cornea after the nodule is excised.
Pterygium excision surgery involves removal of both the corneal and conjunctival components of the lesion. To help decrease the risk of recurrence, meticulous excision of the underlying tenons is important, followed by placement of either an amniotic membrane or conjunctival autograft over the excision site.
As in the case of EBMD, the cornea should be given at least 6 weeks to 8 weeks for the astigmatism to normalize before biometry for IOL calculation is obtained.
Recurrence is a concern following treatment of both SND and pterygium. In the case of pterygium, the recurrence is often more aggressive than the original lesion.
IOL Options
When determining the appropriate IOL for a patient who has irregular astigmatism, the corneal surgeon should weigh the patient’s goals of spectacle and contact lens independence versus corneal regularity. This is so the best lens can be chosen for visual performance, while managing the patient’s postoperative expectations.
The causes of irregular astigmatism will affect the choice of IOL as follows:
• EBMD, SND, or pterygium. If the irregular corneal astigmatism appears more regular on topography after treatment and the magnitude and axis are consistent across multiple devices (topography, tomography, optical biometry, and keratometry), it is reasonable to implant a toric IOL.
Additionally, presbyopia-correcting IOLs and extended-depth-of-focus (EDOF) IOLs are options, depending on the reliability and regularity of corneal astigmatism measurements.
A caveat: The patient should be educated that all the above conditions can recur in the future, with resultant changes in corneal astigmatism, so they are not disappointed postoperatively, should any of these things occur.
In the setting of potentially inaccurate corneal astigmatism measurements, as well as shifting corneal astigmatism, a small aperture IOL is an intriguing option for EBMD, SND, and pterygium. This technology, by using a 1.36 mm central aperture, can create up to 3 D of extended depth of focus, while correcting up to 1.5 D of astigmatism, and tolerating about 1 D of a refractive target miss. The on-label use of this IOL is placement in a patient’s non-dominant eye to achieve increased depth of focus. The success of this IOL in irregular corneas has yet to be determined, as experience with it is needed.
• KCN. IOL power selection in KCN is challenging. Several factors contribute to this: Keratometry measurements are less accurate, effective lens position calculation has increased error, the corneal curvature is not constant along a given meridian, the corneal apex is often off the visual axis and, in general, the corneal power is overestimated in KCN patients. As a result, all the IOL calculation formulas we use tend to result in hyperopic errors. The SRK/T is the most accurate of the older generation formulas, likely because the SRK/T tends toward myopic prediction errors in normal eyes. Modifications have been done to new-generation formulas to further help with accuracy. The Kane Holladay 2, and the Barrett True-K formulas have all shown varying levels of IOL prediction error, but all are still consistently less accurate compared to IOL calculations in non-keratoconus eyes.3
Given the difficulty in IOL power calculation in KCN, implanting diffractive optics or extended-depth-of-focus IOLs may not yield the best results, as their success is contingent on hitting the refractive target. Additionally, the increased aberration, both negative spherical and higher order, can decrease quality of vision further, strengthening the argument against using these IOLs in these patients.
Toric IOLs should be reserved for patients who have mild KCN that has fairly regular astigmatism. The ideal KCN patient has good (relative to the patient) spectacle-corrected visual acuity prior to the development of cataracts and zero desire to wear contact lenses postoperatively.
In KCN, a patient’s best vision will be obtained with a hard contact lens after cataract surgery. If a patient is happy in their hard contact lens and wants to have their best possible vision after surgery, they will need to go back into a hard contact lens. In this case, a toric IOL would actually be contraindicated, as the rigid contact lens nullifies the corneal astigmatism. The goal of a toric IOL in KCN cases is to reduce total refractive error but not necessarily eliminate it; this should be clearly explained to the patient. A zero-aberration IOL, as opposed to an IOL that has negative spherical aberration, may be better suited in KCN patients. This is because the KCN cornea typically has negative spherical aberration in contrast with a healthy cornea that typically has positive spherical aberration.
Finally, a small aperture IOL may be an option in patients who have mild KCN that is centrally located in the apex of the cornea. In patients who have worse KCN, the magnitude of corneal astigmatism may surpass what can be meaningfully corrected with a small aperture IOL. The implications of a displaced corneal apex not being aligned with the centrally located aperture on the IOL is also unclear.
Achieving Patient Satisfaction
When managing patients who have both irregular corneal astigmatism and cataracts, corneal surgeons must diagnose and, if possible, treat the condition before cataract surgery to create satisfied postoperative patients. If the cornea can be rehabilitated to where the astigmatism is fairly regular, a toric IOL can potentially be used. Presbyopia-correcting IOLs may not achieve a desirable result, especially in KCN patients in whom the refractive outcome is uncertain. Time will tell whether the small aperture IOL may be an option for quality of vision and refractive outcomes. CP
References:
- Sridhar MS, Rapuano CJ, Cosar CB, Cohen EJ, Laibson PR. Phototherapeutic keratectomy versus diamond burr polishing of Bowman’s membrane in the treatment of recurrent corneal erosions associated with anterior basement membrane dystrophy. Ophthalmology. 2002;109(4):674-679.
- Nattis AS, Rosenberg ED, Donnenfeld ED. One-year visual and astigmatic outcomes of keratoconus patients following sequential crosslinking and topography-guided surface ablation: the TOPOLINK study. J Cataract Refract Surg. 2020;46(4):507-516.
- Kozhaya K, Chen AJ, Joshi M, et al. Comparison of keratoconus specific to standard IOL formulas in patients with keratoconus undergoing cataract surgery. J Refract Surg. 2023;39(4):242-248.
- Nau CB, Harthan J, Shorter E, et al. Demographic characteristics and prescribing patterns of scleral contact lens fitters: The SCOPE study. Eye Contact Lens. 2018; 44: S265-72.









