Here, I discuss the available options and when to consider each.
Topical/Pharmacologic Interventions
These work best for newly sprouted, immature vessels. Two main treatments are available:
- Corticosteroids. These provide potent anti-inflammatory effects. CNV often requires aggressive topical corticosteroid treatment (e.g., initially every 1 hour) for successful regression.When to prescribe. When inflammation is the primary mechanism of the CNV, controlling inflammation is the best treatment. Inflammatory conditions associated with CNV include herpes simplex virus, immune stromal keratitis, or other forms of resolved infectious keratitis, such as bacterial and fungal keratitis. As the side effects of potential cataract formation and ocular hypertension limit long-term use, topical corticosteroids should be tapered but very slowly (over the course of months) based on clinical response.
- Anti-VEGF agents. Topical, anti-VEGF drugs (i.e., bevacizumab and ranibizumab) inhibit VEGF, precluding new blood vessel formation through down regulation of endothelial cell proliferation.1Reported adverse events include superficial punctate keratitis, epithelial defect formation and persistence, stromal thinning, and eyelid swelling/chalazion.2,3When to prescribe. Corticosteroids are often first-line, as they are the easiest to trial for a potential response to corticosteroids. When the CNV fails to respond, it is reasonable to consider an anti-VEGF agent. Logistically, it may be easier to obtain and administer subconjunctival anti-VEGF, as the topical formulation may only be available at certain compounding pharmacies. Treatment efficacy can be inconsistent, with certain eyes attaining significant CNV reduction and possible elimination long-term, while other eyes can have either no response to treatment or recurrence and persistence of CNV after treatment cessation. Most comparative studies have demonstrated subconjunctival administration was more or equally as effective than topical (less penetration through intact epithelium).4,5 Deeper CNV (and more mature vessels) may be better treated with intrastromal administration, while subconjunctival administration can be better for superficial and mid-stromal vessels.6,7 I often combine topical corticosteroids with anti-VEGF treatment to maximize the anti-inflammatory and antiangiogenic effects.
Of note: Combining anti-VEGF agents, steroids, and an antibiotic has been shown to be more effective in curbing CNV versus anti-VEGF agents alone.1
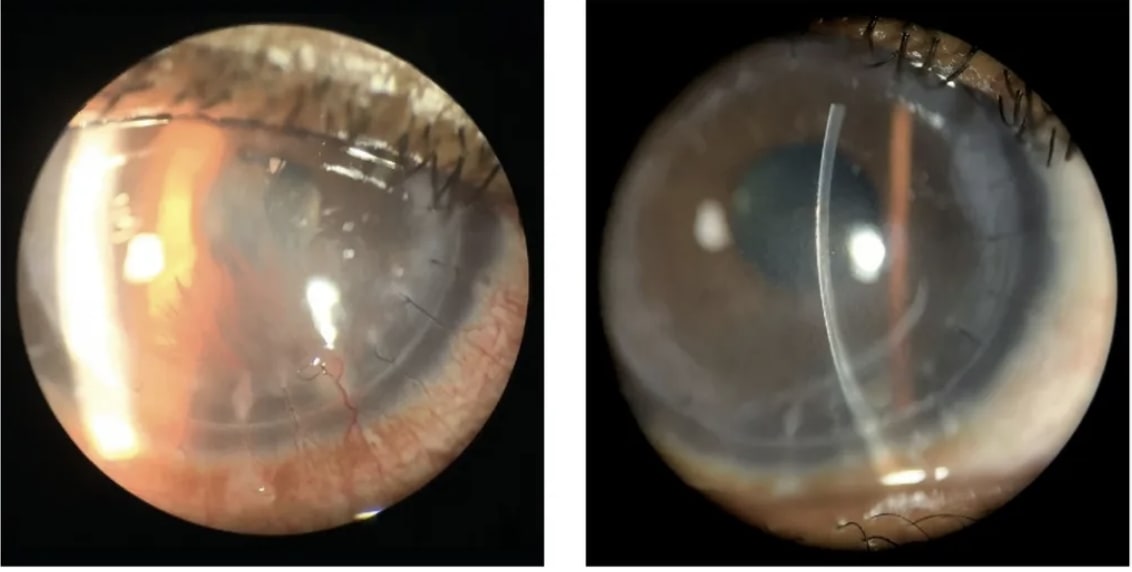
Occlusive Surgical Strategies
For older, more established CNV, vessel disruption is necessary. The following are options:
- Laser thermal cauterization. This can be accomplished using argon, yellow dye, and Nd:YAG lasers. Laser thermal cauterization is often combined with anti-VEGF agents due to the upregulation of VEGF from the inflammatory response of heat-induced coagulation and vessel occlusion. Reported adverse events include iris atrophy, pupil ectasia, corneal hemorrhage, corneal thinning, vascularization exacerbation, and vessel lumen reopening.2Laser choice is typically determined by which laser the provider has. In my experience, argon laser can safely treat vessels at all levels of stroma, as well as small and large caliber vessels; however, treatment through dense stromal haze can be difficult.
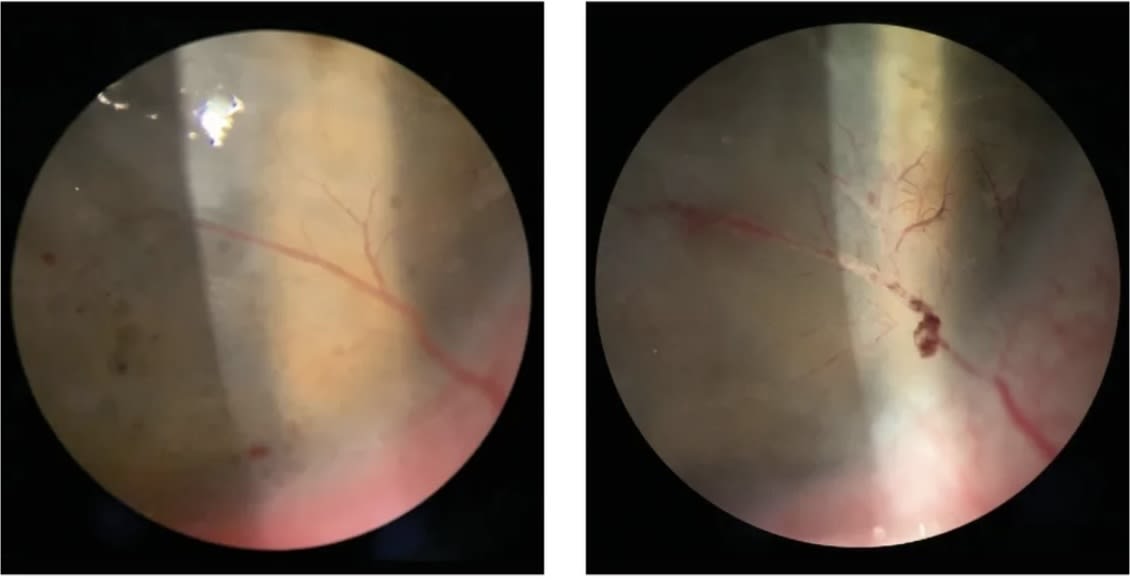
- Fine needle diathermy (FND). This is the use of a coagulating current from a monopolar cautery unit to occlude the afferent (and sometimes efferent) blood vessels. The current is usually delivered through a needle, such as a cutting or electrolysis needle. Adverse events may include intracorneal hemorrhages, microperforations, epithelial defects, and corneal thinning.8When to prescribe. Treating larger caliber vessels can be easier to target with the needle. In one study of 17 eyes, FND decreased corneal vascularization, the amount of lipid deposition, and graft rejection episodes in >80% of eyes that had lipid keratopathy associated with CNV.8 Since FND upregulates VEGF, FND is often combined with anti-VEGF agents to reduce CNV recurrence rates.
- Photodynamic therapy (PDT). PDT with verteporfin involves an intravenous infusion of verteporfin followed by light activation with a nonthermal diode laser to cause vascular photothrombosis of abnormal, leaky CNV vessels. Decreased CNV and evidence of vascular thrombosis was noted in 2/3 of eyes in a case series of 33 eyes.9 Less extensive (fewer number of vessels) and more superficial CNV was more responsive to PDT in this study. Multiple sessions may be necessary to maintain the effect. Cost (especially with multiple sessions) and availability (in an anterior segment practice) of verteporfin may be limiting. Potential adverse events may be systemic (injection site reactions, photosensitivity reactions, and infusion-related back pain) or local (conjunctival hyperemia, superficial hemorrhages, and transient anterior chamber reaction).10
- Mitomycin intravascular chemoembolization (MICE). This involves injection of minute amounts of 0.4 mg/mL mitomycin (0.01 mL to 0.05 mL) into CNV vessels near the limbus with enough retrograde hydrostatic force to fill both the efferent and afferent vessels.11 This technique is able to successfully eliminate the vascular network at times. No local or systemic adverse events have been reported; intrastromal hemorrhage (“pizza pie” sign) was an expected sign from the trapped blood and lipid.When to prescribe. Larger caliber vessels are ideal to target, especially superficial and mid-stromal vessels. Deep vessels may take more experience, given the potential for serious effects if mitomycin is injected into the anterior chamber. Researchers noted complete resolution of high-velocity CNV and subsequent partial absorption of lipid keratopathy.11
- Cryotherapy. This technique has been reported to treat extensive, superficial and intrastromal CNV. A single freeze-thaw cycle of cryotherapy is applied directly to all the areas of the involved cornea. Although this is an easily performed, minimally invasive treatment, there may be damage induced to the corneal endothelial cells.12
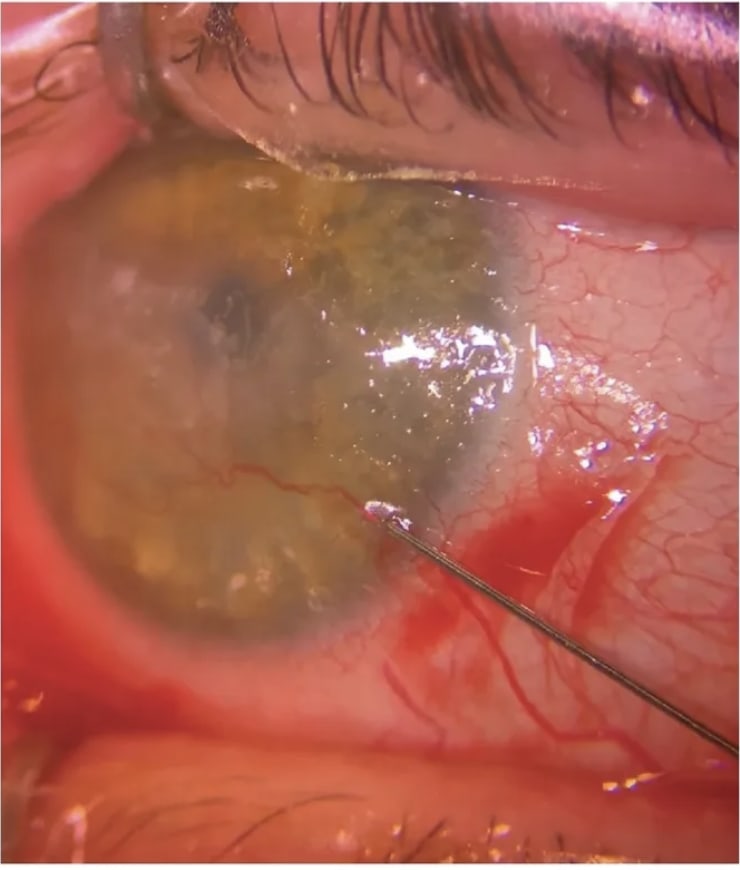
Treatment for Underlying Etiology
For certain CNV etiologies, treating the underlying cause requires a tailored approach (and not standard occlusive strategies). If there is an offending agent, such as suture-inducing neovascularization, management would involve prompt removal.
For contact lens-related CNV from hypoxia, discontinuation of the contact lens is necessary.
Additionally, CNV related to limbal stem cell deficiency can be successfully treated with an ocular surface stem cell transplantation. Certain inflammatory conditions that can have associated CNV (e.g., peripheral ulcerative keratitis, Mooren’s ulcer) may respond to conjunctival resection.
CNV: AN OVERVIEW
CNV is characterized by the infiltration of new blood vessels into the cornea from the limbus. Specifically, these vessels can invade the stroma due to a disruption in the balance of angiogenic and antiangiogenic factors that normally preserve corneal transparency.1
The most common etiologies of CNV include corneal infections (e.g., bacterial/fungal keratitis, herpes simplex virus [HSV], herpes zoster virus), contact lens-induced hypoxia, ocular surface inflammation/injury (e.g., neurotrophic keratitis, limbal stem cell deficiency [LSCD], atopic, blepharitis/meibomian gland dysfunction/rosacea), and graft rejection.2
Most CNV etiologies induce inflammation, leading to an interplay of inflammatory cells (e.g., macrophages, neutrophils) and angiogenic growth factors (i.e., VEGF). Additionally, hypoxia brings about angiogenesis via VEGF expression by cornea epithelial, corneal endothelial, and limbal vessel endothelial cells.2 Further, a relationship exists between corneal innervation and angiogenesis, as denervation induces angiogenesis due, in part, to a decrease in the normal constitutively expressed angiostatic molecules.15
Topography/tomography can illustrate irregularity and higher-order aberrations. Anterior segment angiography can be useful in identifying the afferent/efferent vessels.
Keratoplasty
Addressing corneal neovascularization prior to corneal transplantation can increase the success of the graft. Specifically, in a study of high-risk keratoplasties, 2 mg of preoperative subconjunctival anti-VEGF was administered as monotherapy or with laser coagulation. Over 24.5 months (mean) follow-up, rejection occurred in 23% of the monotherapy group, 35% of the combined therapy group, and 60% of the control group (no antiangiogenic therapy).13 Additionally, in combining a subconjunctival anti-VEGF with FND for eyes with CNV prior to keratoplasty, the estimated probabilities of graft survival rate were 92.9%, 78.4%, and 78.4% for 1, 2, and 3 years, respectively (survival rates comparable to normal-risk keratoplasty eyes).14
Inhibiting Blood Vessel Growth
To preserve optimal vision and safeguard the cornea from structural damage, the cornea’s transparency and avascularity must be maintained. Knowledge of the treatments available for CNV and when, specifically, to prescribe them enable the corneal physician to accomplish this. CP
References:
- Sharif Z, Sharif W. Corneal neovascularization: updates on pathophysiology, investigations & management. Rom J Ophthalmol. 2019;63(1):15-22.
- Roshandel D, Eslani M, Baradaran-Rafii A, et al. Current and emerging therapies for corneal neovascularization. Ocul Surf. 2018;16(4):398-414.
- Kim SW, Ha BJ, Kim EK. The effect of topical bevacizumab on corneal neovascularization. Ophthalmology. 2008;115(6):e33-e38.
- Bhatti N, Qidwai U, Hussain M, Kazi A. Efficacy of sub-conjunctival and topical bevacizumab in high-risk corneal transplant survival. J Pak Med Assoc. 2013 Oct;63(10):1256-9. PMID: 24392555.
- Krizova D, Vokrojova M, Liehneova K, Studeny P. Treatment of Corneal Neovascularization Using Anti-VEGF Bevacizumab. J Ophthalmol. 2014;2014:178132.
- Sarah B, Ibtissam H, Mohammed B, Hasna S, Abdeljalil M. Intrastromal Injection of Bevacizumab in the Management of Corneal Neovascularization: About 25 Eyes. J Ophthalmol. 2016;2016:6084270. doi: 10.1155/2016/6084270.
- S. Maddula, D. K. Davis, S. Maddula, M. K. Burrow, and B. K. Ambati, “Horizons in therapy for corneal angiogenesis,” Ophthalmology, vol. 118, no. 3, pp. 591–599, 2011.
- Sharif Z, Sharif W. Corneal neovascularization: updates on pathophysiology, investigations & management. Rom J Ophthalmol. 2019 Jan-Mar; 63(1): 15–22.
- Faraj LA, Elalfy MS, Said DG, Dua HS. Fine needle diathermy occlusion of corneal vessels. Br J Ophthalmol. 2014;98(9):1287-1290.
- Al-Torbak AA. Photodynamic therapy with verteporfin for corneal neovascularization. Middle East Afr J Ophthalmol. 2012;19(2):185-189.
- Maurizio Fossarello 1, Enrico Peiretti, Ignazio Zucca, Antonina Serra. Photodynamic therapy of corneal neovascularization with verteporfin. Cornea. 2003 Jul;22(5):485-8.
- John TTJ. Cryotherapy offers transplantation alternative for extensive neovascularization, thin corneas. Ocular Surgery News. July 18, 2012. Accessed October 3, 2023. https://www.healio.com/news/ophthalmology/20120725/cryotherapy-offers-transplantation-alternative-for-extensive-neovascularization-thin-corneas
- Trufanov SV, Malozhen SA, Krakhmaleva DA, Surnina ZV, Pivin EA, Kasparova EA. Antiangiogenic therapy in high-risk keratoplasty. Vestn Oftalmol. 2020;136(4):11-18.
- Hos D, Le VNH, Hellmich M, et al. Risk of corneal graft rejection after high-risk keratoplasty following fine-needle vessel coagulation of corneal neovascularization combined with bevacizumab: a pilot study. Transplant Direct. 2019;5(5):e452.
- Ferrari G, Hajrasouliha AR, Sadrai Z, et al. Nerves and neovessels inhibit each other in the cornea. Invest Ophthalmol Vis Sci. 2013;54(1):813-820.









